Посттравматическое стрессовое расстройство: от нейронных сетей к омикс-механизмам и персонализированной терапии
10 марта 2026
Посттравматическое стрессовое расстройство: от нейронных сетей к омикс-механизмам и персонализированной терапии
- 268
- 0
- 2
рисунок автора
-
Автор
-
Редакторы
Статья на конкурс «Био/Мол/Текст»: Посттравматическое стрессовое расстройство представляет собой сложное мультифакторное состояние, формирование и течение которого определяются взаимодействием нейронных, молекулярных и системных механизмов. В обзоре рассматриваются современные представления о патофизиологии ПТСР — от нарушений функционирования нейронных сетей, включающих миндалину, гиппокамп и медиальную префронтальную кору, до изменений на уровне транскриптома, протеома и эпигенетической регуляции. Особое внимание уделено роли метилирования ДНК и микроРНК в формировании устойчивых изменений стресс-реактивности, нейрональной пластичности и угашения страха. Представлены данные о связи эпигенетических изменений с ответом на психотерапевтические и фармакологические вмешательства, включая экспозиционную терапию, EMDR, кетамин и MDMA-assisted therapy. Обсуждаются ограничения существующих исследований, связанные преимущественно с использованием периферических образцов, а также перспективы применения эпигенетических маркеров для стратификации пациентов и развития персонализированных подходов к терапии ПТСР.
Конкурс «Био/Мол/Текст»-2025/2026
Эта работа опубликована в номинации «Свободная тема» конкурса «Био/Мол/Текст»-2025/2026.

Генеральный партнер конкурса — международная инновационная биотехнологическая компания BIOCAD.

Партнер номинации — компания SkyGen: передовой дистрибьютор продукции для life science на российском рынке.

«Книжный» спонсор конкурса — «Альпина нон-фикшн»
Введение
Посттравматическое стрессовое расстройство (ПТСР) — это психическое состояние, которое возникает после переживания травматического события (потеря близких, серьезные аварии, боевые действия, катастрофы, эпидемии и т. д.). У большинства людей острая стрессовая реакция постепенно угасает, однако у 20–30% формируется нарушение переработки травматического опыта, приводящее к развитию ПТСР [1].
По данным международного исследования Всемирной организации здравоохранения, более 70% людей переживают хотя бы одно потенциально травмирующее событие в течение жизни, а у 3–4% развиваются симптомы ПТСР, соответствующие диагностическим критериям [2].
Число людей, подвергающихся травматическому воздействию, остается стабильно высоким. Согласно данным Uppsala Conflict Data Program, после 2010 года наблюдается устойчивый рост числа активных вооруженных конфликтов, а в 2022–2023 гг. их количество достигло максимальных значений за весь период наблюдений, что сопровождается ростом числа гражданских лиц, сталкивающихся с насилием, вынужденным перемещением и утратой близких [3]. Эти факторы напрямую ассоциированы с повышенным риском развития ПТСР [4].
Клиническая картина ПТСР включает навязчивые воспоминания и флэшбеки, избегание стимулов, связанных с травмой, хроническую гипервозбудимость, нарушения сна и концентрации внимания, а также устойчивые негативные изменения настроения и самооценки [5]. Эти симптомы приводят к выраженному снижению трудоспособности, социальной активности и качества жизни.
ПТСР родителей затрагивает не только самих родителей, но и их детей и ближайшее окружение. Мета-анализ показал, что тяжесть симптомов ПТСР у родителей положительно коррелирует с родительским стрессом, степенью конфликтов с детьми и строгостью дисциплины, а также отрицательно с родительской функциональностью. В семьях с травмированными родителями дети чаще испытывают психологический дистресс и поведенческие проблемы, включая тревожность, депрессию и эмоциональную нестабильность [6].
Отдельное значение имеют половые различия: согласно научным обзорам, женщины имеют примерно в два раза более высокий риск развития ПТСР, чем мужчины, даже при сопоставимом уровне травматического воздействия [7]. Это подчеркивает необходимость дальнейшего изучения механизмов ПТСР и разработки дифференцированных профилактических и терапевтических подходов.
Изменения в нейронных-сетях, связанные с развитием ПТСР
Главный фактор развития посттравматического стрессового расстройства заключается не в самом событии, а в том, как мозг и организм закрепляют его в памяти. В норме гиппокамп, миндалина и медиальная префронтальная кора работают согласованно: они соотносят поступающие сигналы с контекстом и регулируют силу эмоциональных реакций (рис. 1).
Гиппокамп отслеживает пространственно-временные характеристики ситуации — где находится человек, что происходило раньше и соответствует ли текущее окружение месту, где действительно возникала угроза. Он передает эту контекстную информацию в миндалину и медиальную префронтальную кору [8]. Опираясь на данные гиппокампа, префронтальная кора определяет, насколько опасна текущая ситуация. Когда она получает сигнал, что контекст отличается от опасного, она усиливает тормозное влияние на центральное ядро миндалины [9].
Миндалина при этом выступает как детектор значимости, который решает, запускать ли тревожную реакцию. Она интегрирует два вида информации: сенсорные сигналы, указывающие на потенциальную угрозу, и контекстные сигналы, позволяющие понять, имеет ли этот сенсорный стимул значение в текущей обстановке. Если гиппокамп и префронтальная кора передают согласованный сигнал безопасности, активность миндалины остается в нормальных пределах. Но при совпадении сенсорного сигнала с контекстом, ранее связанным с опасностью, миндалина активируется и запускает вегетативный стресс-ответ [10]. Центральное ядро миндалины передает сигналы в гипоталамус, который включает симпатическую нервную систему: учащается сердцебиение, повышается давление, дыхание становится поверхностным и быстрым, зрачки расширяются. Через паравентрикулярное ядро гипоталамуса далее активируется HPA-ось — выделяются КРГ (кортико-релизинг гормон), затем АКТГ (адено-кортикотропный гормон) и кортизол [11].
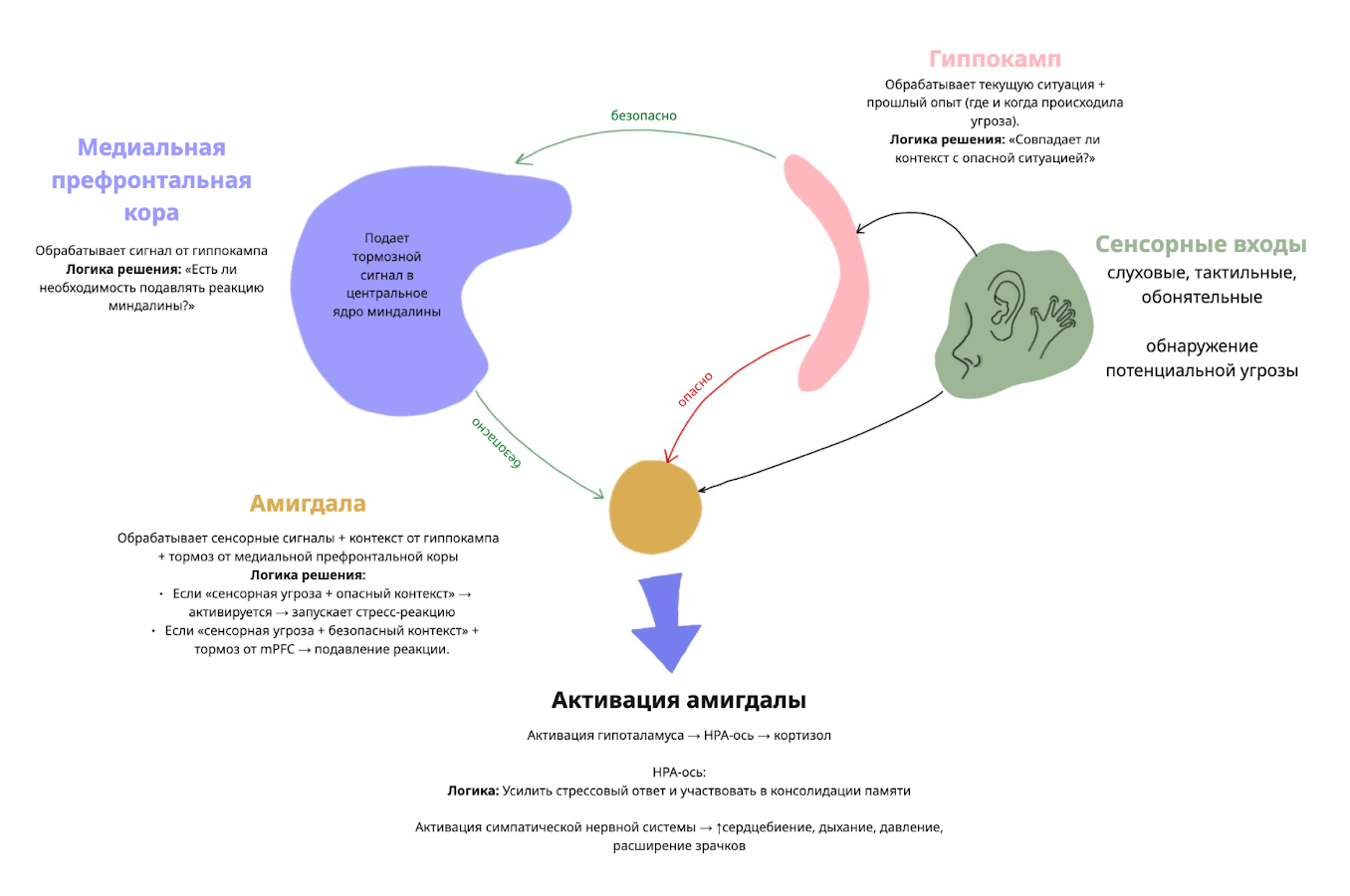
Рисунок 1. Схема взаимодействия миндалины, гиппокампа и префронтальной коры при обработке угроз. Миндалина интегрирует сенсорные и контекстные сигналы и при выявлении опасности активирует вегетативные реакции через гипоталамус и HPA-ось.
рисунок автора
При ПТСР у гиппокампа наблюдаются как структурные, так и метаболические изменения. Исследования демонстрируют уменьшение его объема [12–15] и снижение уровня N-ацетиласпартата (NAA) [16], [17]. Эти показатели отражают ухудшение нейрональной целостности и функционального состояния ткани гиппокампа, что, в свою очередь, связано с нарушениями контекстной памяти. Мозг хуже различает, происходит ли что-то опасное сейчас или это просто напоминание о прошлом. На этом фоне миндалина начинает реагировать без корректного контекстного сигнала, она воспринимает любые похожие стимулы как угрозу. Параллельно сниженная функция медиальной префронтальной коры ухудшает торможение страха — безопасные сигналы больше не гасят реакцию миндалины. В итоге даже нейтральные события вызывают полноценный стресс-ответ [18].
Эти нейронные нарушения приводят к ключевой проблеме — нарушенному угасанию страха.
Для нормального угасания мозг должен сформировать новую, «безопасную» ассоциацию, в которой стимул больше не связан с угрозой. Но если гиппокамп плохо различает контекст, миндалина гиперреактивна, а префронтальная кора не подавляет ее активность, то обучение безопасности становится крайне неэффективным. Поэтому страховые реакции сохраняются и легко возобновляются — даже спустя годы после травмы [19].
Дополняя эти изменения, дисрегуляция HPA-оси поддерживает состояние повышенной настороженности и физиологического напряжения. На таком фоне стресс еще больше усиливает консолидацию травматических воспоминаний, что способствует закреплению симптомов [20].
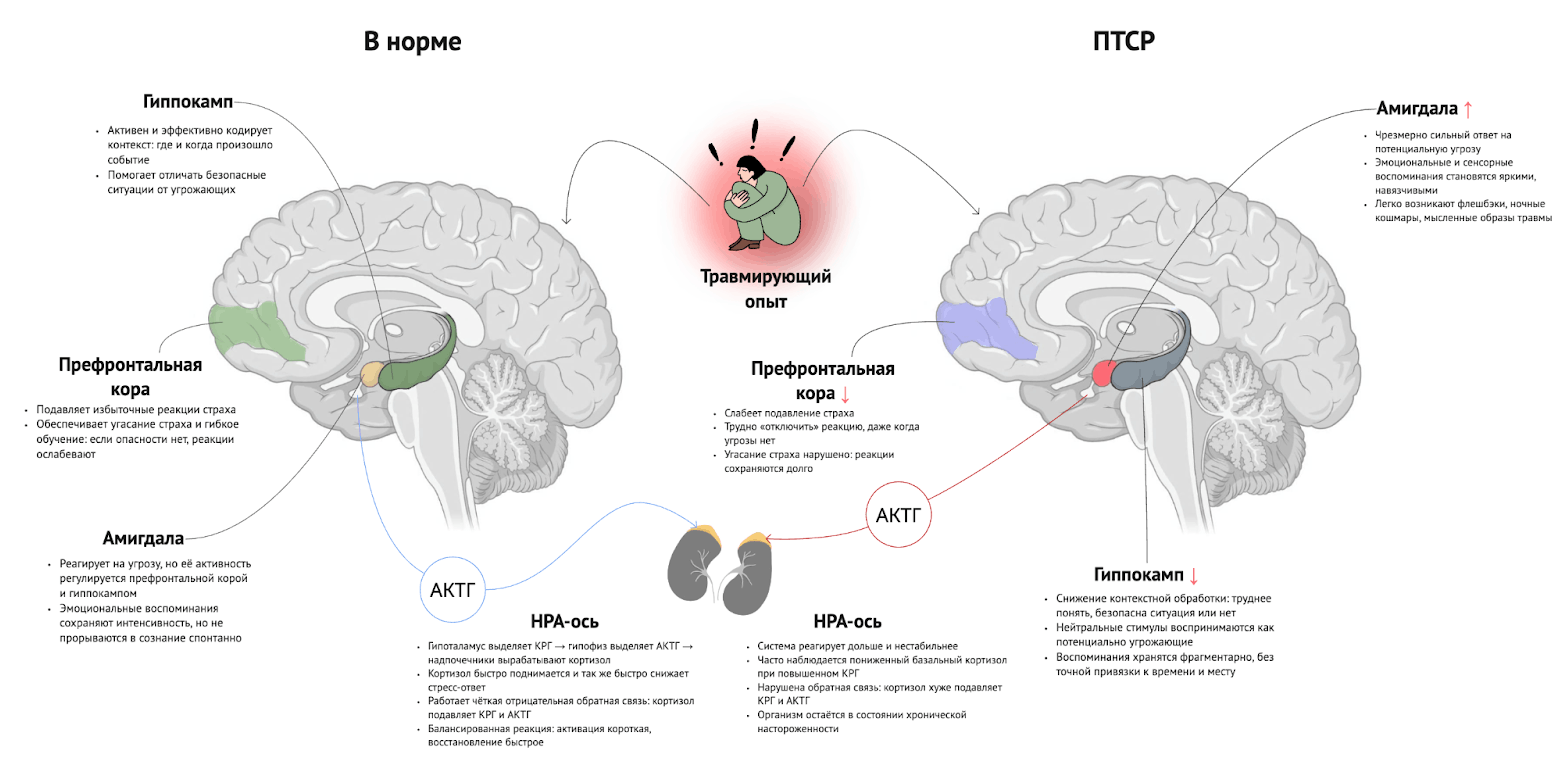
Рисунок 2. Нейронные и нейроэндокринные механизмы обработки травматического опыта в норме и при посттравматическом стрессовом расстройстве (ПТСР).
рисунок автора
Молекулярные изменения, связанные с развитием ПТСР
Применение омиксных подходов к ПТСР показало, что это расстройство затрагивает не только функцию нейронных цепей гиппокамп-медиальная префронтальная кора-миндалина, но и сопровождается глубокой молекулярной перестройкой. При ПТСР изменяется экспрессия генов, паттерны метилирования ДНК, количество и состав белков, метаболитов и липидов, что указывает на комплексное влияние травматического опыта на мозг и организм.
Анализ протеома при ПТСР показал, что происходит уменьшение количества ключевых белков, обеспечивающих ГАМК-ергическую передачу, включая транспортер SLC32A1 [21], который переносит ГАМК — главный тормозной нейромедиатор мозга. ГАМК позволяет нейронам подавлять чрезмерную активность друг друга и контролировать реакции страха и тревоги [22, 23]. Снижение уровня этих белков, включая SLC32A1, может быть связано с ослаблением тормозного контроля со стороны префронтальной коры, что потенциально способствует недостаточной регуляции активности миндалины и усилению тревожных реакций.
Параллельно обнаружено повышение уровня miRNA hsa‑miR‑589 [21], которая может дополнительно блокировать синтез этих белков на посттранскрипционном уровне, в результате даже при нормальной экспрессии генов количество функциональных белков для ГАМК-передачи дополнительно снижается (рис. 3).
В результате комбинации изменений в экспрессии генов и регуляции miRNA, происходит снижение белков-транспортеров при ПТСР что ведет к ослаблению тормозных механизмов и нарушению контроля страха.
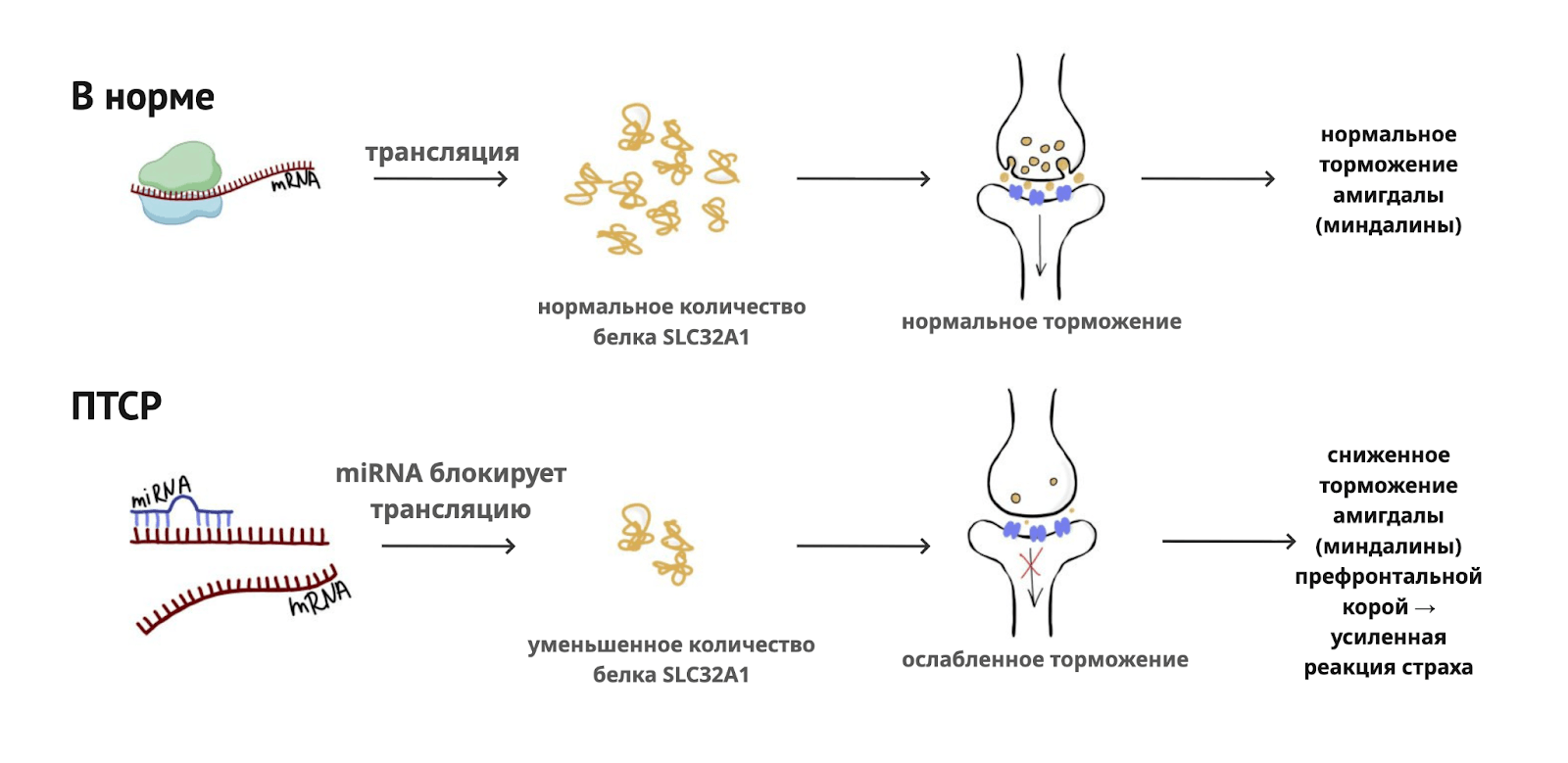
Рисунок 3. Схема процесса синтеза белка (трансляции) без участия микроРНК при синтезе SLC32A1 (в норме), и при участии микроРНК hsa‑miR‑589, что приводит к снижению его уровня.
рисунок автора
Также при ПТСР выявлены изменения метилирования ДНК в регуляторных областях генов, связанных с уязвимостью к стрессу. Метилирование ДНК — это эпигенетический механизм, который регулирует активность генов без изменения их последовательности и обычно приводит к снижению их экспрессии (рис. 4).
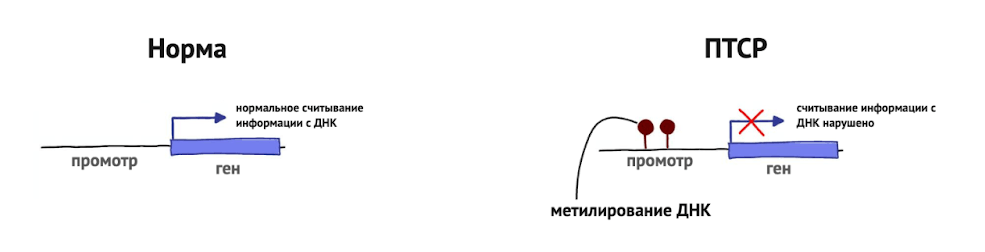
Рисунок 4. Схема процессов синтеза иРНК (транскрипции) без метильной группы (слева) и при наличии метильной метки (справа).
рисунок автора
Среди генов, в регуляторных областях которых выявлены такие эпигенетические изменения, находятся MAD1L1, ELFN1, WNT5A и FKBP5 — гены, участвующие в контроле нейрональной пластичности, синаптической передачи и регуляции стресс-ответа. Изменение их активности может вносить вклад в формирование устойчивых нарушений обработки страха и стресса, характерных для ПТСР.
Например, нарушение экспрессии MAD1L1 [24], [25] может быть связано с изменениями стабильности микротрубочек, что потенциально влияет на транспорт синаптических белков (рис. 5) и может ограничивать эффективность обновления синаптических структур, вовлеченных в процессы угашения страха.

Рисунок 5. Схематическое изображение роли микротрубочек в транспорте рецепторов и белков к синапсам и влияния нарушений экспрессии MAD1L1 на обновление синаптических структур и нейрональную пластичность.
рисунок автора
Изменения метилирования в области ELFN1 [24], [25] отражают нарушения молекулярной регуляции взаимодействия между возбуждающими и тормозными нейронами. Белок ELFN1 экспрессируется в ГАМК‑эргических интернейронах, в частности в интернейронах, содержащих соматостатин, гиппокампа и коры, и локализуется на постсинаптической мембране этих нейронов, где он формирует транс‑синаптическое взаимодействие с метаботропным глутаматным рецептором mGluR7 на пресинаптических терминалях возбуждающих пирамидных нейронов [26]. Это взаимодействие помогает направлять рецепторы mGluR7 на нужные синапсы и контролировать, как часто нейроны передают сигнал. Благодаря этому поддерживается правильный баланс между возбуждением и торможением в цепях нейронов, что важно для контроля активности сети и подавления страха [27], [28]. При нарушении эпигенетической регуляции ELFN1 такая транс-синаптическая модуляция ослабевает, что может способствовать гипервозбудимости нейронных ансамблей и снижению эффективности тормозного контроля. В результате повышается реактивность на угрозу и затрудняется угашение страха (рис. 6).

Рисунок 6. Нарушение транс-синаптического взаимодействия между ГАМК-ергическими интернейронами и возбуждающими нейронами при дисрегуляции ELFN1 и ослаблении mGluR7-зависимой модуляции синаптической передачи.
рисунок автора
Также происходит снижение активности WNT5A [25]. WNT5A — сигнальный белок семейства Wnt — играет ключевую роль в морфогенезе дендритных шипиков и синаптической пластичности. Эксперименты на культурах нейронов и в гиппокампе показывают, что WNT5A стимулирует образование новых шипиков, повышает их плотность и усиливает функции глутаматергических синапсов, что обеспечивает нормальное обучение и формирование долговременной памяти [29], [30].
Кроме того, WNT5A, выделяемый микроглией, способствует созреванию и стабилизации дендритных шипиков, увеличивает электрическую активность нейронов и укрепление синапсов [31]. Снижение активности WNT5A ассоциируется с уменьшением плотности дендритных шипиков (рис. 7) в экспериментальных моделях, что может ограничивать синаптическую пластичность и формирование новых безопасных ассоциаций.
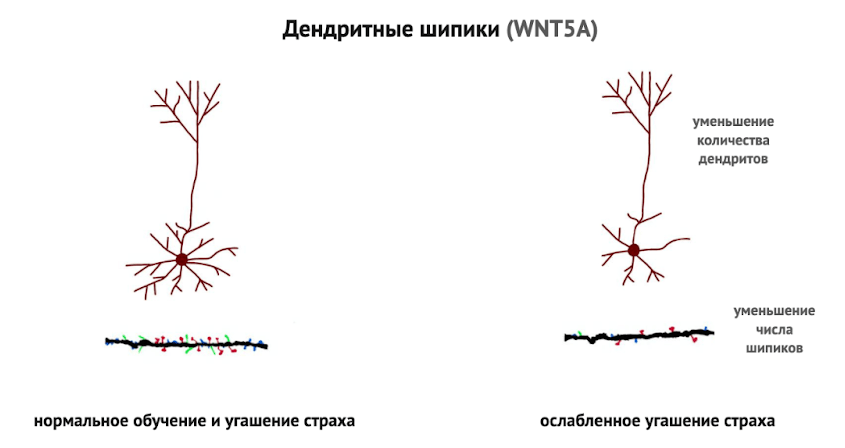
Рисунок 7. Снижение плотности дендритных шипиков и нарушение синаптической пластичности при снижении активности сигнального пути WNT5A.
[32], рисунок адаптирован
Изменения метилирования вокруг FKBP5 [24] нарушают регуляцию чувствительности рецепторов к кортизолу — главному гормону стресса. Ген FKBP5 кодирует белок FKBP51, который регулирует работу глюкокортикоидных рецепторов, участвующих в отрицательной обратной связи HPA‑оси. FKBP51 является кошапероном глюкокортикоидного рецептора и ослабляет активность этого рецептора, снижая способность кортизола эффективно взаимодействовать с ним и подавлять стресс‑ответ [33], [34].
При изменениях метилирования FKBP5 экспрессия FKBP51 может увеличиваться, и из‑за этого глюкокортикоидные рецепторы становятся менее чувствительными к кортизолу, что приводит к удлинению и усилению реакции HPA‑оси на стресс [33], [35].
В норме после кратковременного стрессового воздействия повышенный уровень кортизола должен снижаться через механизм негативной обратной связи, помогая организму вернуться в состояние покоя. Однако из‑за повышенной активности FKBP51 этот механизм нарушается: клетки дольше реагируют на кортизол (рис. 8), а система стресса остается активной дольше [34], [35]. Такая затяжная активация HPA-оси может способствовать более выраженной консолидации стресс-ассоциированных воспоминаний и поддержанию повышенной стресс-реактивности.
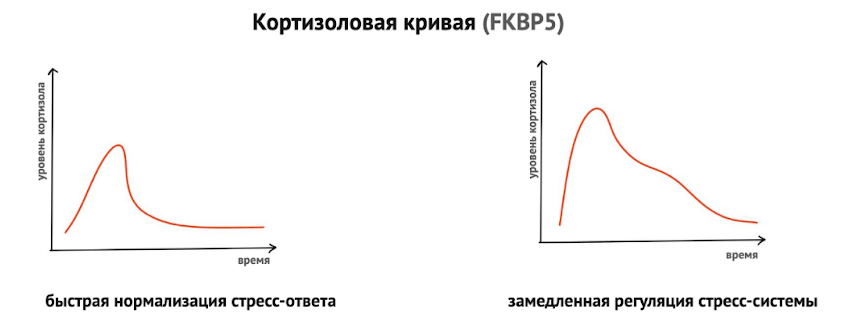
Рисунок 8. Нарушение отрицательной обратной связи гипоталамо-гипофизарно-надпочечниковой оси при повышенной экспрессии FKBP51 и сниженной чувствительности глюкокортикоидных рецепторов к кортизолу.
рисунок автора
Помимо нейронов, при ПТСР страдают глиальные клетки — в особенности микроглия. Поскольку микроглия регулирует воспаление, пластичность и удаление ненужных синапсов, ее подавленная активность означает, что мозг хуже адаптируется после стресса. В областях, отвечающих за контроль эмоций, это приводит к накоплению неэффективных или чрезмерно активных синаптических связей, что дополнительно закрепляет патологические страховые реакции [36].
Персонализированная терапия
Несмотря на наличие различных психотерапевтических и фармакологических подходов к лечению посттравматического стрессового расстройства, клиническая эффективность терапии остается ограниченной. Даже при использовании современных методов, включая экспозиционную терапию и десенсибилизацию и переработку движением глаз (EMDR), клинически значимое снижение симптомов наблюдается лишь у 46–60% пациентов [37]. Такая вариабельность терапевтического ответа указывает на выраженные индивидуальные различия между пациентами и подчеркивает необходимость поиска биологических механизмов, потенциально связанных с чувствительностью к лечению.
Одним из таких механизмов является метилирование ДНК — эпигенетическая модификация, при которой метильные группы присоединяются к цитозину в CpG-локусах и участвуют в регуляции экспрессии генов без изменения нуклеотидной последовательности ДНК [38]. Существенной особенностью метилирования является его чувствительность к факторам окружающей среды, включая стрессовые воздействия, что позволяет рассматривать эпигенетические изменения как потенциально динамический механизм, посредством которого внешние воздействия могут приводить к устойчивым молекулярным изменениям [39], [40].
Существуют данные, что метилирование ДНК может изменяться под воздействием внешних факторов, включая терапевтические вмешательства [41], [42]. В исследованиях, посвященных ПТСР, показано, что успешное лечение может сопровождаться изменениями метилирования ДНК, в частности в генах, участвующих в регуляции стресс-ответа. Показано, что клиническое улучшение может ассоциироваться с частичным обращением ранее выявленных эпигенетических изменений в крови [43]. В крупных клинических исследованиях также отмечено, что у пациентов с выраженным снижением интенсивности симптомов после психотерапии наблюдались изменения метилирования CpG-локуса cg25535999 гена NR3C1, тогда как при слабом терапевтическом ответе такие изменения выражались значительно меньше [44]. Пилотные данные дополнительно указывают, что изменения метилирования в генах NR3C1, FKBP5 и CRHR1 могут коррелировать с клиническим улучшением, однако эти результаты остаются предварительными и требуют подтверждения в более крупных выборках [45].
В то же время ряд исследований не выявил значимых терапевтически ассоциированных изменений метилирования у части пациентов [46]. Это подчеркивает, что эпигенетический ответ на лечение является неоднородным, и один и тот же терапевтический метод может сопровождаться различными молекулярными эффектами у разных людей.
Важно, что в совокупности эти данные указывают не только на то, что эпигенетические метки способны изменяться под влиянием терапии, но и на то, что при различном клиническом ответе наблюдаются различные направления и выраженность этих изменений [44–46]. Это позволяет предположить, что различия в эпигенетическом состоянии могут существовать уже до начала лечения и влиять на индивидуальную чувствительность к терапии. Поэтому изучение метилирования ДНК является перспективным подходом для понимания того, какие биологические механизмы лежат в основе различий терапевтического ответа, и в дальнейшем может способствовать более обоснованному выбору лечебных стратегий для конкретного пациента. Однако большинство данных получено из исследований крови [47–50], тогда как эпигенетические процессы имеют выраженную тканевую и клеточную специфичность [25], поэтому периферические маркеры не всегда отражают изменения в мозге [40]. Несмотря на эти ограничения, накопленные результаты свидетельствуют о значительном потенциале дальнейшего изучения эпигенетических механизмов в контексте персонализированного подхода к терапии ПТСР.
Выводы
Современные данные свидетельствуют о том, что ПТСР не может рассматриваться исключительно как следствие дисфункции отдельных мозговых структур или нейромедиаторных систем. Нарушения в согласованной работе гиппокампа, миндалины и медиальной префронтальной коры формируют основу патологической переработки травматического опыта, однако устойчивость симптомов определяется более глубокими молекулярными изменениями, затрагивающими регуляцию экспрессии генов, эпигенетические механизмы и нейрональную пластичность. Изменения метилирования ДНК и регуляции микроРНК, затрагивающие гены, связанные с контролем стресс-ответа, синаптической передачи и структурной перестройки нейронных сетей, могут способствовать закреплению нарушений угашения страха и гиперреактивности к угрозе.
В то же время данные указывают на потенциальную обратимость части эпигенетических изменений при эффективном лечении, что открывает перспективы использования молекулярных маркеров для прогнозирования терапевтического ответа. Несмотря на то что большинство исследований основано на анализе образцов крови и не всегда напрямую отражает процессы, происходящие в мозге, согласованность результатов различных работ делает эпигенетические механизмы обоснованным и перспективным направлением дальнейших исследований. В долгосрочной перспективе интеграция нейронных, омиксных и клинических данных может создать основу для развития персонализированных подходов к терапии ПТСР, потенциально способствующих более устойчивому терапевтическому ответу и снижению вероятности длительного сохранения симптомов.
Литература
- Tadesse Tarik Tamir, Berhan Tekeba, Enyew Getaneh Mekonen, Deresse Abebe Gebrehana, Alebachew Ferede Zegeye. (2025). Shadows of trauma: an umbrella review of the prevalence and risk factors of post-traumatic stress disorder in children and adolescents. Child Adolesc Psychiatry Ment Health. 19;
- Ronald C. Kessler, Sergio Aguilar-Gaxiola, Jordi Alonso, Corina Benjet, Evelyn J. Bromet, et. al.. (2017). Trauma and PTSD in the WHO World Mental Health Surveys. European Journal of Psychotraumatology. 8;
- Rustad S.A. (2024) Conflict Trends: A Global Overview, 1946–2023. PRIO Paper;
- Fiona Charlson, Mark van Ommeren, Abraham Flaxman, Joseph Cornett, Harvey Whiteford, Shekhar Saxena. (2019). New WHO prevalence estimates of mental disorders in conflict settings: a systematic review and meta-analysis. The Lancet. 394, 240-248;
- American Psychiatric Association Diagnostic and Statistical Manual of Mental Disorders — American Psychiatric Association, 2013;
- Jessica E. Lambert, Jessica Holzer, Amber Hasbun. (2014). Association Between Parents’ PTSD Severity and Children's Psychological Distress: A Meta‐Analysis. Journal of Traumatic Stress. 27, 9-17;
- David F. Tolin, Edna B. Foa. (2006). Sex differences in trauma and posttraumatic stress disorder: A quantitative review of 25 years of research.. Psychological Bulletin. 132, 959-992;
- Ahmed A. Moustafa, Mark W. Gilbertson, Scott P. Orr, Mohammad M. Herzallah, Richard J. Servatius, Catherine E. Myers. (2013). A model of amygdala–hippocampal–prefrontal interaction in fear conditioning and extinction in animals. Brain and Cognition. 81, 29-43;
- Fredrik Åhs, Philip A. Kragel, David J. Zielinski, Rachael Brady, Kevin S. LaBar. (2015). Medial prefrontal pathways for the contextual regulation of extinguished fear in humans. NeuroImage. 122, 262-271;
- Young-Rae Kim, Moonhyung Lee, Man S. Kim. (2025). Astrocytes in Fear Memory Processing: Molecular Mechanisms Across the Amygdala–Hippocampus–Prefrontal Cortex Network. Cells. 14, 1444;
- Lisa M Shin, Israel Liberzon. (2010). The Neurocircuitry of Fear, Stress, and Anxiety Disorders. Neuropsychopharmacol. 35, 169-191;
- . (1995). MRI-based measurement of hippocampal volume in patients with combat- related posttraumatic stress disorder. AJP. 152, 973-981;
- Tamara V. Gurvits, Martha E. Shenton, Hiroto Hokama, Hirokazu Ohta, Natasha B. Lasko, et. al.. (1996). Magnetic resonance imaging study of hippocampal volume in chronic, combat-related posttraumatic stress disorder. Biological Psychiatry. 40, 1091-1099;
- M. B. STEIN, C. KOVEROLA, C. HANNA, M. G. TORCHIA, B. McCLARTY. (1997). Hippocampal volume in women victimized by childhood sexual abuse. Psychol. Med.. 27, 951-959;
- Gerardo Villarreal, Derek A Hamilton, Helen Petropoulos, Ira Driscoll, Laura M Rowland, et. al.. (2002). Reduced hippocampal volume and total white matter volume in posttraumatic stress disorder. Biological Psychiatry. 52, 119-125;
- Thomas W. Freeman, David Cardwell, Craig N. Karson, Richard A. Komoroski. (1998). In vivo proton magnetic resonance spectroscopy of the medial temporal lobes of subjects with combat‐related posttraumatic stress disorder. Magnetic Resonance in Med. 40, 66-71;
- Gerardo Villarreal, Helen Petropoulos, Derek A Hamilton, Laura M Rowland, William P Horan, et. al.. (2002). Proton Magnetic Resonance Spectroscopy of the Hippocampus and Occipital White Matter in PTSD: Preliminary Results. Can J Psychiatry. 47, 666-670;
- Kerry. J. Ressler, Sabina Berretta, Vadim Y. Bolshakov, Isabelle M. Rosso, Edward G. Meloni, et. al.. (2022). Post-traumatic stress disorder: clinical and translational neuroscience from cells to circuits. Nat Rev Neurol. 18, 273-288;
- Mohammed R. Milad, Gregory J. Quirk. (2012). Fear Extinction as a Model for Translational Neuroscience: Ten Years of Progress. Annu. Rev. Psychol.. 63, 129-151;
- Yehuda R. (2001). Biology of posttraumatic stress disorder. J. Clin. Psychiatry. 62, 41–46;
- Jiawei Wang, Yujing Liu, Hongyu Li, Tuan P. Nguyen, John Lee Soto-Vargas, et. al.. (2025). A multi-omic approach implicates novel protein dysregulation in post-traumatic stress disorder. Genome Med. 17;
- Olga Babaev, Carolina Piletti Chatain, Dilja Krueger-Burg. (2018). Inhibition in the amygdala anxiety circuitry. Exp Mol Med. 50, 1-16;
- Lydiard R.B. (2003). The role of GABA in anxiety disorders. J. Clin. Psychiatry. 64, 21–27;
- Ahyeon Hwang, Mario Skarica, Siwei Xu, Jensine Coudriet, Che Yu Lee, et. al.. (2025). Single-cell transcriptomic and chromatin dynamics of the human brain in PTSD. Nature. 643, 744-754;
- Hongyu Li, Yujing Liu, Yiming Shi, Jiawei Wang, Tuan P. Nguyen, et. al.. (2025). Mapping DNA Methylation Signatures to Identify Epigenetic Variation Across Subcortical Regions of the Human Posttraumatic Stress Disorder Brain. Biological Psychiatry;
- Naoko H. Tomioka, Hiroki Yasuda, Hiroyuki Miyamoto, Minoru Hatayama, Naoko Morimura, et. al.. (2014). Elfn1 recruits presynaptic mGluR7 in trans and its loss results in seizures. Nat Commun. 5;
- Tevye Jason Stachniak, Emily Lauren Sylwestrak, Peter Scheiffele, Benjamin J. Hall, Anirvan Ghosh. (2019). Elfn1-Induced Constitutive Activation of mGluR7 Determines Frequency-Dependent Recruitment of Somatostatin Interneurons. J. Neurosci.. 39, 4461-4474;
- Hayato Matsunaga, Jun Aruga. (2021). Trans-Synaptic Regulation of Metabotropic Glutamate Receptors by Elfn Proteins in Health and Disease. Front. Neural Circuits. 15;
- Lorena Varela-Nallar, Iván E. Alfaro, Felipe G. Serrano, Jorge Parodi, Nibaldo C. Inestrosa. (2010). Wingless-type family member 5A (Wnt-5a) stimulates synaptic differentiation and function of glutamatergic synapses. Proc. Natl. Acad. Sci. U.S.A.. 107, 21164-21169;
- Eva Ramos-Fernández, Macarena S. Arrázola, Carolina A. Oliva, Sebastián B. Arredondo, Lorena Varela-Nallar, Nibaldo C. Inestrosa. (2021). Wnt5a promotes hippocampal postsynaptic development and GluN2B-induced expression via the eIF2α HRI kinase. Sci Rep. 11;
- Hana Yeh, Maya E. Woodbury, Kaitlin L. Ingraham Dixie, Tsuneya Ikezu, Seiko Ikezu. (2023). Microglial WNT5A supports dendritic spines maturation and neuronal firing. Brain, Behavior, and Immunity. 107, 403-413;
- Xinzhao Chen, Yifan Jiang, Jiayu Wang, Yishu Liu, Menglei Xiao, et. al.. (2020). Synapse impairment associated with enhanced apoptosis in post‐traumatic stress disorder. Synapse. 74;
- Jonathan J. Sabbagh, Ricardo A. Cordova, Dali Zheng, Marangelie Criado-Marrero, Andrea Lemus, et. al.. (2018). Targeting the FKBP51/GR/Hsp90 Complex to Identify Functionally Relevant Treatments for Depression and PTSD. ACS Chem. Biol.. 13, 2288-2299;
- Alexander S. Häusl, Lea M. Brix, Jakob Hartmann, Max L. Pöhlmann, Juan-Pablo Lopez, et. al.. (2021). The co-chaperone Fkbp5 shapes the acute stress response in the paraventricular nucleus of the hypothalamus of male mice. Mol Psychiatry. 26, 3060-3076;
- Anthony S Zannas, Tobias Wiechmann, Nils C Gassen, Elisabeth B Binder. (2016). Gene–Stress–Epigenetic Regulation of FKBP5: Clinical and Translational Implications. Neuropsychopharmacol. 41, 261-274;
- Jing Zhang, Alfred P. Kaye, Jiawei Wang, Matthew J. Girgenti. (2021). Transcriptomics of the depressed and PTSD brain. Neurobiology of Stress. 15, 100408;
- Julie Rendum Klaeth, Andreas Gjerde Jensen, Trude Julie Brynhildsvoll Auren, Stian Solem. (2024). 12-month follow-up of intensive outpatient treatment for PTSD combining prolonged exposure therapy, EMDR and physical activity. BMC Psychiatry. 24;
- Lisa D Moore, Thuc Le, Guoping Fan. (2013). DNA Methylation and Its Basic Function. Neuropsychopharmacol. 38, 23-38;
- Kevin J. Dudley, Xiang Li, Michael S. Kobor, Tod E. Kippin, Timothy W. Bredy. (2011). Epigenetic mechanisms mediating vulnerability and resilience to psychiatric disorders. Neuroscience & Biobehavioral Reviews. 35, 1544-1551;
- Divya Mehta, Olivia Miller, Dagmar Bruenig, Georgina David, Jane Shakespeare‐Finch. (2020). A Systematic Review of DNA Methylation and Gene Expression Studies in Posttraumatic Stress Disorder, Posttraumatic Growth, and Resilience. Journal of Traumatic Stress. 33, 171-180;
- Jacob Peedicayil. (2011). Epigenetic management of major psychosis. Clin Epigenet. 2, 249-256;
- Lap Ho, Patricia A. Bloom, Joan G. Vega, Shrishailam Yemul, Wei Zhao, et. al.. (2016). Biomarkers of Resilience in Stress Reduction for Caregivers of Alzheimer’s Patients. Neuromol Med. 18, 177-189;
- Christiaan H. Vinkers, Elbert Geuze, Sanne J. H. van Rooij, Mitzy Kennis, Remmelt R. Schür, et. al.. (2021). Successful treatment of post-traumatic stress disorder reverses DNA methylation marks. Mol Psychiatry. 26, 1264-1271;
- Sarah Wilker, Vanja Vukojevic, Anna Schneider, Anett Pfeiffer, Stefan Inerle, et. al.. (2023). Epigenetics of traumatic stress: The association of NR3C1 methylation and posttraumatic stress disorder symptom changes in response to narrative exposure therapy. Transl Psychiatry. 13;
- Candace R. Lewis, Joseph Tafur, Sophie Spencer, Joseph M. Green, Charlotte Harrison, et. al.. (2023). Pilot study suggests DNA methylation of the glucocorticoid receptor gene (NR3C1) is associated with MDMA-assisted therapy treatment response for severe PTSD. Front. Psychiatry. 14;
- Elisabeth Hummel, Magdeldin Elgizouli, Maurizio Sicorello, Elsa Leitão, Jasmin Beygo, et. al.. (2022). No evidence for intervention-associated DNA methylation changes in monocytes of patients with posttraumatic stress disorder. Sci Rep. 12;
- Alicia K. Smith, Karen N. Conneely, Varun Kilaru, Kristina B. Mercer, Tamara E. Weiss, et. al.. (2011). Differential immune system DNA methylation and cytokine regulation in post‐traumatic stress disorder. American J of Med Genetics Pt B. 156, 700-708;
- D. Mehta, D. Bruenig, T. Carrillo‐Roa, B. Lawford, W. Harvey, et. al.. (2017). Genomewide
DNA methylation analysis in combat veterans reveals a novel locus forPTSD . Acta Psychiatr Scand. 136, 493-505; - the Traumatic Stress Brain Study Group, Mark W. Logue, Mark W. Miller, Erika J. Wolf, Bertrand Russ Huber, et. al.. (2020). An epigenome-wide association study of posttraumatic stress disorder in US veterans implicates several new DNA methylation loci. Clin Epigenet. 12;
- B P F Rutten, E Vermetten, C H Vinkers, G Ursini, N P Daskalakis, et. al.. (2018). Longitudinal analyses of the DNA methylome in deployed military servicemen identify susceptibility loci for post-traumatic stress disorder. Mol Psychiatry. 23, 1145-1156.
Комментарии
Раньше здесь был блок с комментариями. Но потом сервис Disqus, на котором они работали и за который мы платили, перестал открываться из РФ.
Когда появится возможность, мы вернём комментарии уже на внутреннем движке, а чтобы это произошло быстрее —
Оставьте донат 💚